Summary
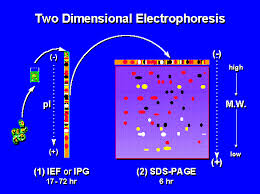
SDS-polyacrylamide gel electrophoresis technology was established in 1967 by Shapire. SDS-PAGE is formed into gel by Acr, crosslinking agent N, and Bis in the presence of the catalyst ammonium persulfate and TEMED, and then this gel can act as support of electrophoresis.
Theory
SDS-PAGE can divide proteins into several pieces of zones according to the difference of charged and different mobility caused by different molecular weight. If there is only one kind of protein, and there will be only one zone after electrophoresis. SDS-PAGE can cover the different charge between protein molecules by increasing the amount of negatively charged protein. So the mobility of SDS protein complexes during electrophoresis will not be affected by the original charge and molecular shape, but by molecular function. This method is called SDS-PAGE technology.
Features
1.At a certain concentration, the gel is transparent, flexible with good mechanical properties.
2.It also has stable chemical properties. There is no chemical reaction with the isolated. And it is also insoluble in many solvents.
3.It is more stable to pH and temperature variations.
4.Almost no adsorption and electro-osmotic effect. The sample separation will have good repeatability, as long as Acr has high purity and the consistent operating conditions.
5.Sample is not easy to spread with less samples and high sensitivity.
6.Gel pore size and concentration can be adjusted according to the molecular weight.
7.It has high resolution especially in discontinuous gel electrophoresis.
Experimental steps:
1.Concentrating effect of the sample
Conventional discontinuous electrophoresis system comprising upper and lower tank buffer (Tris-Gly, pH8.3), stacking gel buffer (Tris-HCl, pH6.8), the separation gel buffer (Tri s-HCl, pH8. 8) has the two different gel concentration. Under these conditions, the HCl buffer system almost all dissociated into Cl -, only a small portion of Gly in two grooves is dissociated into ions, and acidic protein can be also dissociated into ion. These ions move to the cathode at electrophoresis. Cl is followed by protein and Gly anion. Cl will form a zone with low conductance, steep and high potential gradient. This is because of the electrochemical gradient discontinuity, protein and G1y ions accelerate, which is good for improving the resolution of electrophoresis.
2.Molecular sieve effect
Negative effects of Gly will be increased along with the increase of the pH of protein ions. The mobility will be down as the increase of gel concentration. These changes make the movement speed of Gly faster than proteins. And proteins will move in a stable environment of pH and voltage gradient according to its molecular sizes. Pore size of separating gel is different and so that they have different effect on proteins with different molecular size. That is the theory for molecular sieve effect.
Notes:
1.The combination of SDS and proteins should be according to a proportion. Protein content can not be exceeded, or the SDS content is not enough.
2.Standard curve should be made at the same time with SDS-PAGE experiment.
3.Some proteins are composed of the subunit proteins (e.g., hemoglobin) or more than two peptide chains, which will be dissociated into subunits or multiple single peptides at the presence of mercaptoethanol and SDS. So as for this kind of proteins, SDS-PAGE technology can only detect their relative molecular weight.
These are some basic information and experimental steps and notes for SDS-PAGE. Creative Proteomics can offer 2d sds page, as well as the service of sds-page.
SDS-polyacrylamide gel electrophoresis technology was established in 1967 by Shapire. SDS-PAGE is formed into gel by Acr, crosslinking agent N, and Bis in the presence of the catalyst ammonium persulfate and TEMED, and then this gel can act as support of electrophoresis.
Theory
SDS-PAGE can divide proteins into several pieces of zones according to the difference of charged and different mobility caused by different molecular weight. If there is only one kind of protein, and there will be only one zone after electrophoresis. SDS-PAGE can cover the different charge between protein molecules by increasing the amount of negatively charged protein. So the mobility of SDS protein complexes during electrophoresis will not be affected by the original charge and molecular shape, but by molecular function. This method is called SDS-PAGE technology.
Features
1.At a certain concentration, the gel is transparent, flexible with good mechanical properties.
2.It also has stable chemical properties. There is no chemical reaction with the isolated. And it is also insoluble in many solvents.
3.It is more stable to pH and temperature variations.
4.Almost no adsorption and electro-osmotic effect. The sample separation will have good repeatability, as long as Acr has high purity and the consistent operating conditions.
5.Sample is not easy to spread with less samples and high sensitivity.
6.Gel pore size and concentration can be adjusted according to the molecular weight.
7.It has high resolution especially in discontinuous gel electrophoresis.
Experimental steps:
1.Concentrating effect of the sample
Conventional discontinuous electrophoresis system comprising upper and lower tank buffer (Tris-Gly, pH8.3), stacking gel buffer (Tris-HCl, pH6.8), the separation gel buffer (Tri s-HCl, pH8. 8) has the two different gel concentration. Under these conditions, the HCl buffer system almost all dissociated into Cl -, only a small portion of Gly in two grooves is dissociated into ions, and acidic protein can be also dissociated into ion. These ions move to the cathode at electrophoresis. Cl is followed by protein and Gly anion. Cl will form a zone with low conductance, steep and high potential gradient. This is because of the electrochemical gradient discontinuity, protein and G1y ions accelerate, which is good for improving the resolution of electrophoresis.
2.Molecular sieve effect
Negative effects of Gly will be increased along with the increase of the pH of protein ions. The mobility will be down as the increase of gel concentration. These changes make the movement speed of Gly faster than proteins. And proteins will move in a stable environment of pH and voltage gradient according to its molecular sizes. Pore size of separating gel is different and so that they have different effect on proteins with different molecular size. That is the theory for molecular sieve effect.
Notes:
1.The combination of SDS and proteins should be according to a proportion. Protein content can not be exceeded, or the SDS content is not enough.
2.Standard curve should be made at the same time with SDS-PAGE experiment.
3.Some proteins are composed of the subunit proteins (e.g., hemoglobin) or more than two peptide chains, which will be dissociated into subunits or multiple single peptides at the presence of mercaptoethanol and SDS. So as for this kind of proteins, SDS-PAGE technology can only detect their relative molecular weight.
These are some basic information and experimental steps and notes for SDS-PAGE. Creative Proteomics can offer 2d sds page, as well as the service of sds-page.

 RSS Feed
RSS Feed
