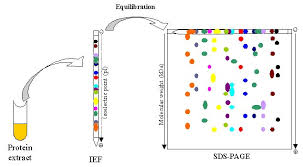
Summary
Once I wrote an article about the experimental theory of 2D-Electrophoresis. Here I want to share some experimental steps for this technology. In my last article, I mainly introduce the theory of IEF and SDS-PAGE without the steps of this experiment.
Before we start this experiment, we should prepare samples, reagents and equipment. As for these tips, we are not going to talk about it. Here we mainly introduce the experimental steps.
Before using the equilibration buffer, we should add 100mg DTT into each 10ml equilibration buffer. Adding an appropriate amount of equilibration buffer I and bromophenol blue solution according to the following statistics. Taking out IPG strips and putting into glass tubes and then we can use the parafilm to cover them. After that, they should be shaken on a shaker for 15 minutes and then pouring equilibration buffer I.
Adding 400mg iodoacetamide (the equilibration buffer II) to each 10ml equilibration buffer. And adding the appropriate amount of equilibration buffer II and bromophenol blue solution according to the table below. Sealing with prafilm, shaking on a shaker for 15 minutes and draining equilibration buffer II.
IPG strips balance fluid volume.
Strip length (cm) recommend balancing fluid volume (ml) / Article bromophenol blue solution (μl)
7 2.5-5 12.5-25
1 15-10 25-50
13 5-10 25-50
18 10 50
24 15 70
Washing IPG strips with deionized water for 1 second and putting the edge strip on filter paper for several minutes and then removing the equilibration buffer.
Transferring IPG strip:
Locating IPG strip on the surface of the glass plate, thereby making the strip support film against one of the glass plate. And then pushing the IPG strip softly with a thin ruler to make a full contact between the IPG strip and plate glass. We should be sure that there are no bubbles.
In SDS-PAGE aspect
Step Solution (250ml per gel) Gel Type
1mm unbacked 1mm on film or glass support or 1.5 unbacked
Fixing: 25ml acetic acid, 100ml methanol 125ml milli-Q water 2x15 min 2x60 min
Sensitization: 75ml methanol, 0.5g Na2S2O3, 17g NaAc, milli-Q water to 250ml 30 min 60 min
Rinsing: 250ml milli-Q water 3x5 min 5x8 min
Silver staining: 0.625g AgNO3, milli-Q water to 250ml 20 min 60 min
Color: 6.25g Na2CO3, 100μl formaldehyde, milli-Q water to 250ml 4 min 6 min
Termination: 3.65g EDTA, milli-Q water to 250 ml 10 min 40 min
Rinsing: 250ml milli-Q water 3x5 min 2x30 min
Silver staining Note:
Many silver staining procedures are using glutaraldehyde, which can improve the sensitivity of silver staining and dyeing reproducibility of results. However, the glutardialdehyde will modify proteins and then the MS detection and analysis of proteins will be affected. Following steps we should pay more attention:
1.Ensuring that all stained containers are absolutely clean and we can use stained glass or plastic containers;
2.Using distilled water (conductivity <2μS) to ensure the staning result.
3.Do not touch the gel with naked hands during dyeing process.
These are main tips for this experiment.
Once I wrote an article about the experimental theory of 2D-Electrophoresis. Here I want to share some experimental steps for this technology. In my last article, I mainly introduce the theory of IEF and SDS-PAGE without the steps of this experiment.
Before we start this experiment, we should prepare samples, reagents and equipment. As for these tips, we are not going to talk about it. Here we mainly introduce the experimental steps.
Before using the equilibration buffer, we should add 100mg DTT into each 10ml equilibration buffer. Adding an appropriate amount of equilibration buffer I and bromophenol blue solution according to the following statistics. Taking out IPG strips and putting into glass tubes and then we can use the parafilm to cover them. After that, they should be shaken on a shaker for 15 minutes and then pouring equilibration buffer I.
Adding 400mg iodoacetamide (the equilibration buffer II) to each 10ml equilibration buffer. And adding the appropriate amount of equilibration buffer II and bromophenol blue solution according to the table below. Sealing with prafilm, shaking on a shaker for 15 minutes and draining equilibration buffer II.
IPG strips balance fluid volume.
Strip length (cm) recommend balancing fluid volume (ml) / Article bromophenol blue solution (μl)
7 2.5-5 12.5-25
1 15-10 25-50
13 5-10 25-50
18 10 50
24 15 70
Washing IPG strips with deionized water for 1 second and putting the edge strip on filter paper for several minutes and then removing the equilibration buffer.
Transferring IPG strip:
Locating IPG strip on the surface of the glass plate, thereby making the strip support film against one of the glass plate. And then pushing the IPG strip softly with a thin ruler to make a full contact between the IPG strip and plate glass. We should be sure that there are no bubbles.
In SDS-PAGE aspect
Step Solution (250ml per gel) Gel Type
1mm unbacked 1mm on film or glass support or 1.5 unbacked
Fixing: 25ml acetic acid, 100ml methanol 125ml milli-Q water 2x15 min 2x60 min
Sensitization: 75ml methanol, 0.5g Na2S2O3, 17g NaAc, milli-Q water to 250ml 30 min 60 min
Rinsing: 250ml milli-Q water 3x5 min 5x8 min
Silver staining: 0.625g AgNO3, milli-Q water to 250ml 20 min 60 min
Color: 6.25g Na2CO3, 100μl formaldehyde, milli-Q water to 250ml 4 min 6 min
Termination: 3.65g EDTA, milli-Q water to 250 ml 10 min 40 min
Rinsing: 250ml milli-Q water 3x5 min 2x30 min
Silver staining Note:
Many silver staining procedures are using glutaraldehyde, which can improve the sensitivity of silver staining and dyeing reproducibility of results. However, the glutardialdehyde will modify proteins and then the MS detection and analysis of proteins will be affected. Following steps we should pay more attention:
1.Ensuring that all stained containers are absolutely clean and we can use stained glass or plastic containers;
2.Using distilled water (conductivity <2μS) to ensure the staning result.
3.Do not touch the gel with naked hands during dyeing process.
These are main tips for this experiment.

 RSS Feed
RSS Feed
