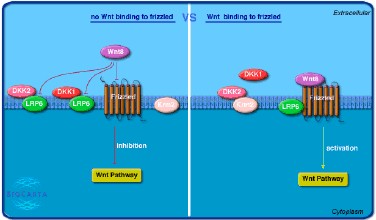
Thyroid hormone (T3) acts on chondrocytes and osteoblasts to control bone development and maintenance, but the pathway in these functions is not clear. Graham R. Williams from Imperial College London found that thyroid hormone receptor mutation (the TRPC) can activate Wnt signaling of bone in vivo and indicated there is interaction between T3 and Wnt signaling pathways.
Since the dominant negative mutant T3 receptor (TRβPV) can not bind T3 to interfere with the function of the wild-type TR, ThrbPV/PV thyroid axis of mice’s pituitary is severely damaged, companied with the elevated thyroid hormone levels. Thrb PV/PV mice accelerate the bone growth through unknown mechanisms. Researchers studied the main osteoblasts from wild-type mice and ThrbPV/PV mice with the help of Wnt array method. Analyzing bone Wnt target genes by in situ hybridization method, which identified that there is activated Wnt signaling during postnatal growth period.
In contrast, dealt with T3, osteoblasts Wnt signaling will be inhibited. This indicates that T3 prevents the accumulation of β- catenin in vivo through promoting the degradation of β- catenin by proteasome, in order to inhibit Wnt signaling pathways. However, as for ThrbPV/PV mice, Wnt signaling pathways are activated by the functional TRβ PV, which stabilizes the β-catenin.
These research illustrates the interaction between T3 and Wnt signaling pathways during the period of regulating bone growth and development.
Since the dominant negative mutant T3 receptor (TRβPV) can not bind T3 to interfere with the function of the wild-type TR, ThrbPV/PV thyroid axis of mice’s pituitary is severely damaged, companied with the elevated thyroid hormone levels. Thrb PV/PV mice accelerate the bone growth through unknown mechanisms. Researchers studied the main osteoblasts from wild-type mice and ThrbPV/PV mice with the help of Wnt array method. Analyzing bone Wnt target genes by in situ hybridization method, which identified that there is activated Wnt signaling during postnatal growth period.
In contrast, dealt with T3, osteoblasts Wnt signaling will be inhibited. This indicates that T3 prevents the accumulation of β- catenin in vivo through promoting the degradation of β- catenin by proteasome, in order to inhibit Wnt signaling pathways. However, as for ThrbPV/PV mice, Wnt signaling pathways are activated by the functional TRβ PV, which stabilizes the β-catenin.
These research illustrates the interaction between T3 and Wnt signaling pathways during the period of regulating bone growth and development.
 RSS Feed
RSS Feed
