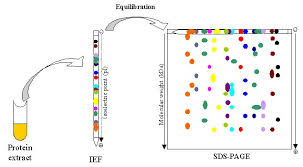
As the increasing needs on biological technologies, especially those used for the protein separation, Scientists developed many technologies, such as mass spectrometry and 2d electrophoresis. This article is mainly about some tips during performing 2d electrophoresis in order to gain the terrific results.
Tips in sample preparation
1.Chemicals, which are applied for all steps in 2d electrophoresis, should be analytical grade quality at least. Storing solutions should always be stored and made at -70°C. During this step, researchers should try to avoid the repeated thawing and freezing of solutions and samples and the reuse of running buffer.
2.Avoiding keratin contamination by using clean and dust-free vessels.
3.Avoiding protein losses with the as simple sample preparation as possible. Dealing with a new sample, two different cell disruption protocols and comparasion of protein yield and qualitative protein content.
Tips in experimental preparation
1.Because of the time-dependent processes, such as denaturation, solubilization and disaggregation of proteins during sample preparation, so it is very important and necessary for researchers to incubate proteins in 2DE lysis buffer for 1h at room temperature.
2.Some substances, such as salt, ionic acids, lipids and so on, can be also great factors that affect the result. These substances should be removed. And if necessary, protease and phosphatase inhibitor cocktails should be added to the 2DE lysis buffer before the cell lysis.
3.It would be better to centrifuge the sample solution before the IEFsample application for 1h at 40,000g, for the insoluble material in the sample solution can block the pores of the IPG strip.
4.Should the urea-containing buffers are heated above 37°C, protein carbmylation may occur.
5.The removal of interfering abundant proteins or nonrelevant classes of protein allow to yield proteins of interest at detected levels.
6.Protein concentration about 5–10 µg/µL will lead a good result.
7.IPG strips have to be rehydrated, being covered, to their original thickness of 0.5mm for 12h at 20°C.
8.The air bubbles should be avoided during the rehydration and the IPG strips should not stick to the bottom of the rehydration tray.
9.Rinsing and blotting IPG gel strips with deionized water after the rehydration, in order to remove the excess rehydration solution.
Tips in experimental process
1.Starting IEF with a low-voltage gradient and limit current to 50 µA per IPG strip, which will get a good result in sample entry.
2.After that, the IPG strips should be stored at -70°C(at least 3-6months) or applied to a second dimension SDS-PAGE gel.
3.There should be no bubbles at the 2nd dimension gel. And sealing the IPG strip in place by using warm agarose.
4.Moving proteins from the IPG strip into the gel and then program the recommended voltage for the remainder of the run with a low voltage.
5.Staining proteins at room temperature with gentle agitation, and without any dust during this process.
6.Reducing the imaging duration if the images are saturated.
These are some tips, which can help researchers gain a good result of the electrophoresis 2d.
Tips in sample preparation
1.Chemicals, which are applied for all steps in 2d electrophoresis, should be analytical grade quality at least. Storing solutions should always be stored and made at -70°C. During this step, researchers should try to avoid the repeated thawing and freezing of solutions and samples and the reuse of running buffer.
2.Avoiding keratin contamination by using clean and dust-free vessels.
3.Avoiding protein losses with the as simple sample preparation as possible. Dealing with a new sample, two different cell disruption protocols and comparasion of protein yield and qualitative protein content.
Tips in experimental preparation
1.Because of the time-dependent processes, such as denaturation, solubilization and disaggregation of proteins during sample preparation, so it is very important and necessary for researchers to incubate proteins in 2DE lysis buffer for 1h at room temperature.
2.Some substances, such as salt, ionic acids, lipids and so on, can be also great factors that affect the result. These substances should be removed. And if necessary, protease and phosphatase inhibitor cocktails should be added to the 2DE lysis buffer before the cell lysis.
3.It would be better to centrifuge the sample solution before the IEFsample application for 1h at 40,000g, for the insoluble material in the sample solution can block the pores of the IPG strip.
4.Should the urea-containing buffers are heated above 37°C, protein carbmylation may occur.
5.The removal of interfering abundant proteins or nonrelevant classes of protein allow to yield proteins of interest at detected levels.
6.Protein concentration about 5–10 µg/µL will lead a good result.
7.IPG strips have to be rehydrated, being covered, to their original thickness of 0.5mm for 12h at 20°C.
8.The air bubbles should be avoided during the rehydration and the IPG strips should not stick to the bottom of the rehydration tray.
9.Rinsing and blotting IPG gel strips with deionized water after the rehydration, in order to remove the excess rehydration solution.
Tips in experimental process
1.Starting IEF with a low-voltage gradient and limit current to 50 µA per IPG strip, which will get a good result in sample entry.
2.After that, the IPG strips should be stored at -70°C(at least 3-6months) or applied to a second dimension SDS-PAGE gel.
3.There should be no bubbles at the 2nd dimension gel. And sealing the IPG strip in place by using warm agarose.
4.Moving proteins from the IPG strip into the gel and then program the recommended voltage for the remainder of the run with a low voltage.
5.Staining proteins at room temperature with gentle agitation, and without any dust during this process.
6.Reducing the imaging duration if the images are saturated.
These are some tips, which can help researchers gain a good result of the electrophoresis 2d.
 RSS Feed
RSS Feed
